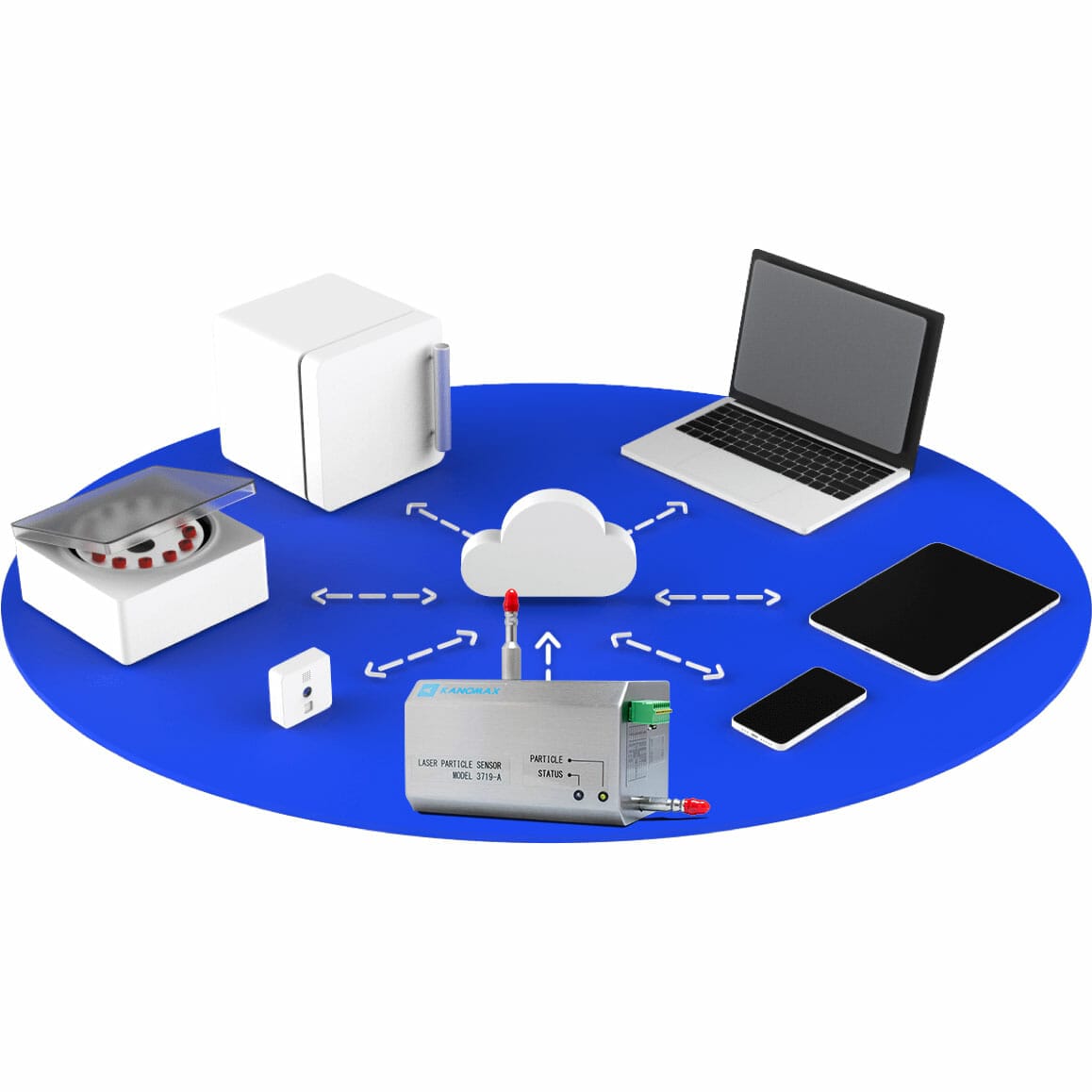
Kanomax has extensive experience with cleanroom contamination control and monitoring solutions.
The Kanomax Cleanroom Monitoring System offers turnkey solutions for monitoring needs required by various industries such as pharmaceutical, medical device, aerospace, semiconductor, and automotive.
Capable of real time monitoring of temperature, humidity, airborne particles, differential pressure, energy consumption, and gases. Local or cloud-based data server available for monitoring data. Initiate alarms per designated event and have dependable remote access. Integrates Elemental Machines technologies.
More Information
Navigate the tab section below to see this product’s Features & Benefits, Specifications, Resources, and more.
- Real time monitoring of temperature, humidity, airborne particles, differential pressure, energy consumption, gases
- Cloud-based data server for monitoring data
- Initiate alarms per event
- Dependable remote access
- Security and compliant results
- System validation
- Meet all the necessary regulatory requirements: 21 CFR Part 11, ISO 14644, USP797/800
- Save money and resources for monitoring system project
- Provides a turnkey solution for a cleanroom monitoring project. Kanomax will help with:
- Developing a system proposal
- Designing monitoring system
- System installation
- System validation
- Training
- Technical support & troubleshooting
- Calibration of sensors
- Kanomax helps customers determine cleanroom monitoring requirements
- A monitoring system can be installed and begin operating within 3 months
- Cleanroom monitoring for Pharmaceutical manufacturing (FDA cGMP and EU GMP)
- Cleanroom monitoring project, which requires formal validation
- Small scale cleanroom startup project (we can offer as small as 1 sensor system)
Examples of previous project types:
- Aerospace
- Advanced Material
- Automotive parts manufacturing
- Electrics assemble
- Medical device manufacturing
- Pharmaceutical packaging
- USP797 compounding pharmacy
Data Sheet - English
Introducing the Kanomax Cleanroom Monitoring System
Learn more about Kanomax USA’s comprehensive and customizable cleanroom monitoring systems, made in partnership with Elemental Machines.
Our sales representatives are here to help you find the right instrument for your application. Fast.
Request a quote today to find the product you need with expert assistance.